Parents are being urged to dispose of dosing syringes found in 14 batches of Panadol Children.
The Therapeutic Goods Administration (TGA) and parent company Haleon Australia issued a product defect correction Panadol Children 1 Month to 1 Year paracetamol 100mg/mL oral liquid bottle, which is used for temporary relief of pain and fever.
“If the dosing syringe is stiff, you may not be able to control the delivery of the medicine,” the TGA said.
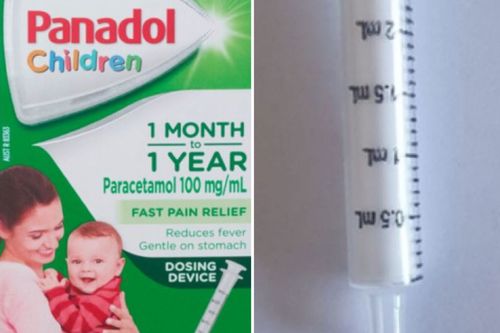
“This poses a risk of choking, especially for babies.
“No other batches of this product, or other Panadol Children products, are affected.
“There are no problems with the liquid medicine itself.”
The affected batch numbers for the 20mL bottle are:
- DR562 (expiry 30/04/2024)
- DR563 (expiry 30/04/2024)
- DT528 (expiry 30/04/2024)
- DS947 (expiry 31/07/2024)
- DS956 (expiry 31/08/2024)
- DT334 (expiry 30/09/2024)
- DT469 (expiry 30/09/2024)
- DT762 (expiry 31/10/2024)
- DT912 (expiry 31/10/2024)
- DU128 (expiry 30/11/2024)
- DT913 (expiry 31/10/2024)
- DW301 (expiry 28/02/2025)
- DW507 (expiry 28/02/2025)
- DW487 (expiry 28/02/2025)
“If you have Panadol Children 1 Month to 1 Year paracetamol 100mg/mL oral liquid bottle, check if it is from the batches listed above,” the TGA said.
“The batch number and expiry date are on the base of the carton and on the label of the bottle. Dispose of the supplied syringe and use an alternative.
“You can seek an alternative syringe from a pharmacy.
“You can contact Haleon Consumer Relations on 1800 028 533 from 8.30am – 5pm weekdays, Eastern Standard Time if you have any questions.
“If you are concerned about using the syringe, please consult your pharmacist.”







